Receiving a cancer diagnosis presents profound difficulties, encompassing both the severe physical symptoms and the considerable emotional burden placed on patients and their loved ones. The path through treatment often requires making complex choices regarding care strategies, each with its own inherent uncertainties and profound implications for daily existence. In this context, many are actively seeking innovative insights that could offer fresh avenues for managing these formidable challenges. This article will delve into a fascinating, evolving research approach that promises to fundamentally change our understanding of how to combat cancer cells, revealing its full potential step by step.
The Foundational Role of Light in Biomedical Research
Light has historically served as a vital instrument across diverse health disciplines, ranging from basic diagnostic procedures to sophisticated therapeutic applications. More recently, scientific investigators have intensified their focus on understanding how particular forms of light interact with biological cells at a molecular scale. This renewed interest is driven by a critical demand for therapeutic strategies that offer high precision while minimizing bodily disruption. Emerging research demonstrates that specific light wavelengths possess the capability to modulate cellular activities without recourse to broadly invasive interventions. Furthermore, a crucial aspect involves effectively leveraging light to precisely target designated areas, integrating insights from both physics and biology.
Unpacking the Science of This Novel Therapeutic Strategy
Central to this groundbreaking methodology is the deployment of near-infrared (NIR) light, renowned for its superior tissue penetration capabilities compared to conventional visible light. Scientists have developed and tested specialized aminocyanine dyes designed to selectively bind to the membranes of cancer cells, rendering them highly sensitive to NIR light exposure. Upon activation, these dyes induce a rapid and intense vibration within the cell membranes, ultimately causing them to destabilize and rupture. Controlled laboratory experiments have demonstrated remarkable efficacy, showing the potential to eliminate up to 99% of targeted cancer cells.
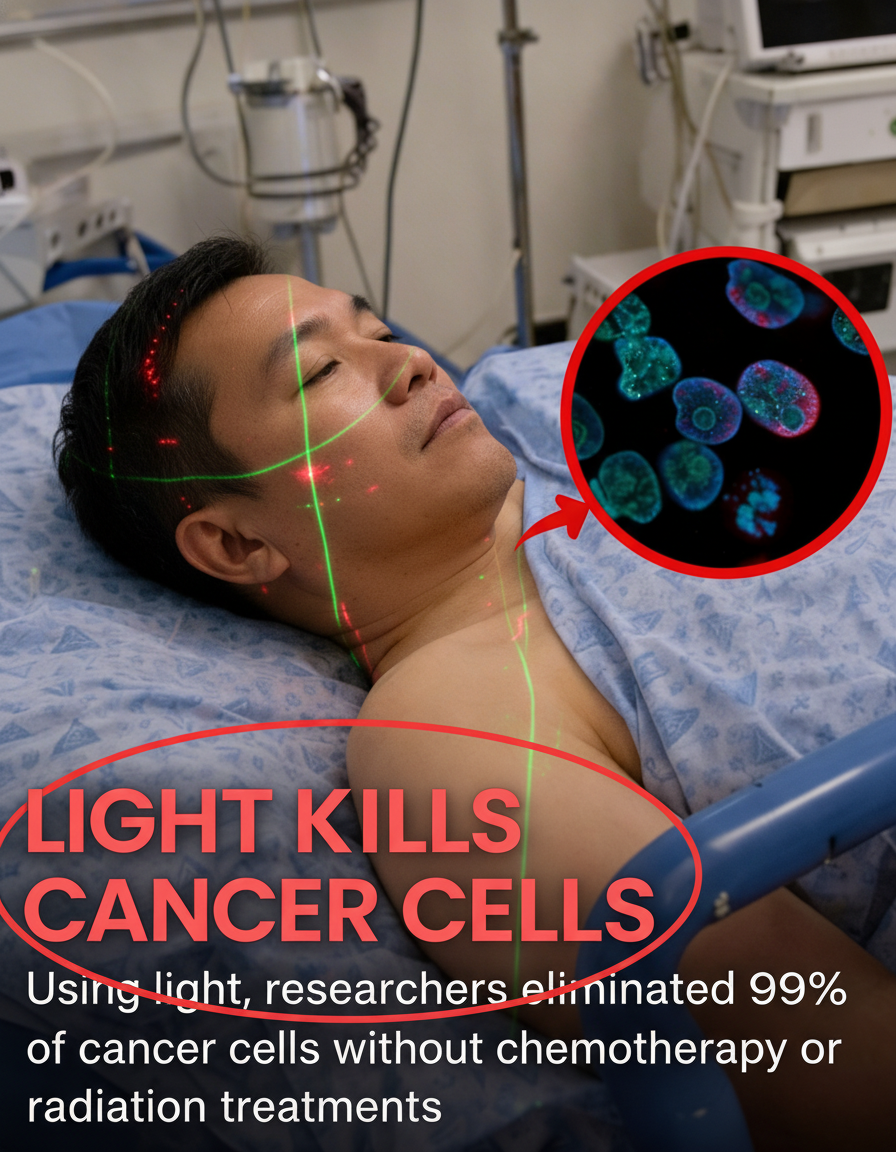
This innovative technique, drawing on research from prominent institutions such as Rice University and Texas A&M, specifically utilizes aminocyanine molecules. These compounds are highly biocompatible and have existing applications in medical imaging, underscoring their safety profile. A particularly compelling aspect of this strategy is its fundamental divergence from conventional cancer treatments: it operates without generating heat or employing harsh chemicals, thereby mitigating potential damage to adjacent healthy tissues.
- Precision Targeting: This therapy ensures activation solely of the specifically dyed cancer cells, leaving healthy surrounding cells unharmed.
- Minimal Invasiveness: The approach bypasses the need for extensive surgical interventions, focusing instead on precise, localized energy delivery.
- Versatility: Initial preclinical investigations indicate a broad applicability of this method across a diverse range of cancer cell types.
Further insights, detailed in a publication in Nature Chemistry, reveal the astonishing speed at which these molecular vibrations occur—exceeding one million cycles per second. This ultra-rapid oscillation is what effectively leads to the mechanical dismantling of the targeted cellular structures.
Promising Early Outcomes from Preclinical Investigations
The initial findings from laboratory experiments are highly encouraging, demonstrating substantial reductions in populations of cultured cancer cells. Notably, melanoma cells exhibited particular sensitivity, responding with impressive efficacy rates. Progressing to in vivo studies using animal models, specifically mice implanted with tumors, this novel therapy induced significant tumor regression without causing observable damage to surrounding healthy tissues. The research team’s experts highlight that while these results draw upon decades of foundational photodynamic research, they introduce a distinct mechanical mechanism. Furthermore, in hypothetical clinical applications, patients could potentially benefit from a reduced side-effect profile, given that this method circumvents systemic impacts.
| Aspect | Conventional Therapies | Light-Activated Approach |
|---|---|---|
| Targeting Mechanism | Generalized exposure | Precise molecular vibration |
| Potential Side Effects | Higher, often due to widespread |